|
Please click on the element name for complete list of element properties. You can sort the elements by clicking on the table headers. List of chemical elements in periodic table with atomic number, chemical symbol and atomic weight. Use this website at any time when you need to get fast and precise information about atomic or proton number of chemical elements. There is also some extra summary on every each chemical element which can be found at our website, including the atomic weight of each element, as well as physical and chemical properties of every element and its importance. By using our website, you can do it in just one click and receive short and correct information on this matter. This website is created for those who need to know the atomic number of a central chemical element. 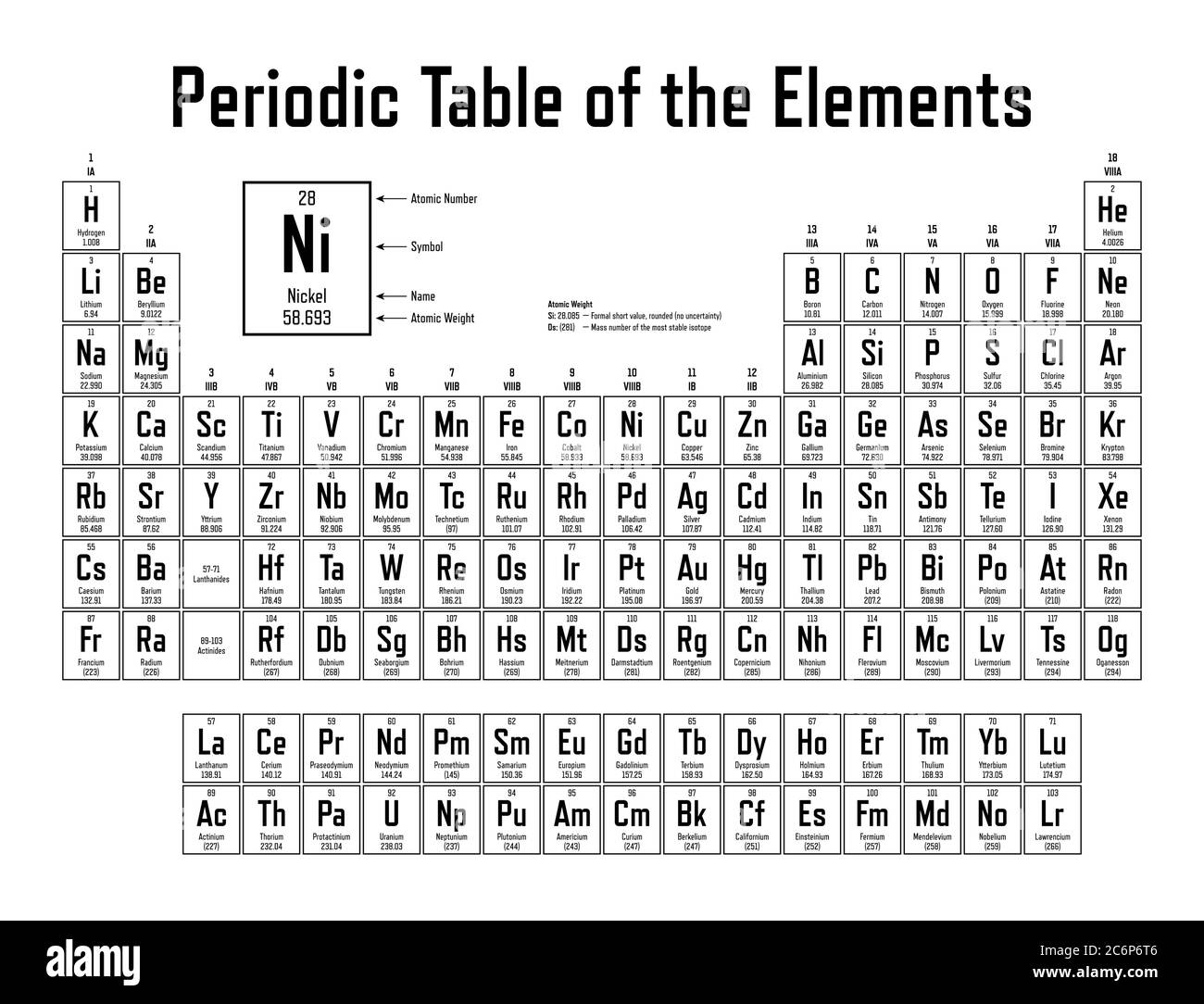
The atomic number is marked with the symbol Z, taken from a German word zahl (or atomzahl, which is ' atomic number' in German). History of Element Names Lots of fun info Periodic Table Dynamic Periodic Table. Atomic number of an element never changes: for example, the atomic number of oxygen is always 8, and the atomic number of Chlorine is always 18. Find Your Name in the Periodic Table of the Elements, create custom pens, save the images. Also, knowing the atomic number of an element can give us an idea about the position of the element in the Periodic Table. In addition to the elements name, symbol, and atomic number, each element box has a drawing of. Why is this so? Why is the atomic number so important? First of all, it is the number that makes elements different from one another as it shows the number of protons in their nuclei. Periodic Table of the Elements, in Pictures and Words. It is an organizing icon of chemistry and is widely used in physics and other sciences. This number can be really important and something essential to know, in relation to a certain chemical element which is the issue of our interest at the moment. Chemistry Portal v t e The periodic table, also known as the periodic table of the elements, arranges the chemical elements into rows ('periods') and columns ('groups'). It is always the whole number and it ranges from 1 to 118, according to the number of the element in the Periodic Table. It is the same as the number of protons that the atom of each element has, so sometimes atomic number is called proton number. We remember from our school chemistry course that every element has its own specific atomic number. /what-are-the-first-20-elements-608820-FINAL-5b758ab446e0fb002c67279a.png) /what-are-the-first-20-elements-608820-FINAL-5b758ab446e0fb002c67279a.png)
Atomic Number of Elements in Periodic Table The Chemistry Division's Periodic Table describes the history, properties, resources, uses, isotopes, forms, costs, and other information for each element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
